Diversity and composition of pelagic prokaryotic and protist communities in a thin Arctic sea-ice regime
Abstract
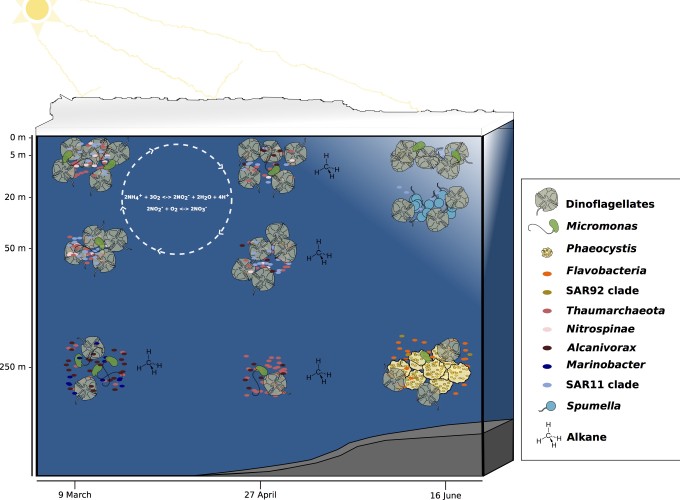
One of the most prominent manifestations of climate change is the changing Arctic sea-ice regime with a reduction in the summer sea-ice extent and a shift from thicker, perennial multiyear ice towards thinner, first-year ice. These changes in the physical environment are likely to impact microbial communities, a key component of Arctic marine food webs and biogeochemical cycles. During the Norwegian young sea ICE expedition (N-ICE2015) north of Svalbard, seawater samples were collected at the surface (5 m), subsurface (20 or 50 m), and mesopelagic (250 m) depths on 9 March, 27 April, and 16 June 2015. In addition, several physical and biogeochemical data were recorded to contextualize the collected microbial communities. Through the massively parallel sequencing of the small subunit ribosomal RNA amplicon and metagenomic data, this work allows studying the Arctic’s microbial community structure during the late winter to early summer transition. Results showed that, at compositional level, Alpha- (30.7%) and Gammaproteobacteria (28.6%) are the most frequent taxa across the prokaryotic N-ICE2015 collection, and also the most phylogenetically diverse. Winter to early summer trends were quite evident since there was a high relative abundance of thaumarchaeotes in the under-ice water column in late winter while this group was nearly absent during early summer. Moreover, the emergence of Flavobacteria and the SAR92 clade in early summer might be associated with the degradation of a spring bloom of Phaeocystis. High relative abundance of hydrocarbonoclastic bacteria, particularly Alcanivorax (54.3%) and Marinobacter (6.3%), was also found. Richness showed different patterns along the depth gradient for prokaryotic (highest at mesopelagic depth) and protistan communities (higher at subsurface depths). The microbial N-ICE2015 collection analyzed in the present study provides comprehensive new knowledge about the pelagic microbiota below drifting Arctic sea-ice. The higher microbial diversity found in late winter/early spring communities reinforces the need to continue with further studies to properly characterize the winter microbial communities under the pack-ice.